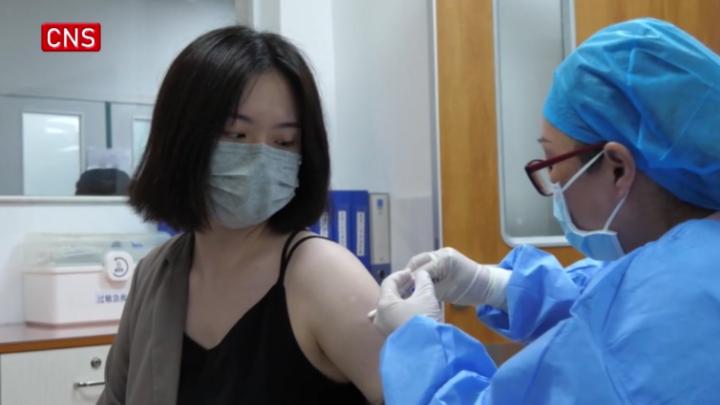
Sinopharm, the Chinese pharmaceutical giant, has commenced phase-1 and phase-2 clinical trials for its new recombinant vaccine against COVID-19 in central China's Henan Province since Saturday.
Developed by the China National Biotec Group (CNBG), a Sinopharm's bioscience subsidiary, the vaccine was issued with a clinical research permit from the National Medical Products Administration on April 9.
This is the CNBG's third COVID-19 vaccine candidate approved for human trials.
Researchers started the early-staged clinical trials in the city of Shangqiu, Henan Province. The trials were randomized, double-blind and placebo-controlled, according to a company statement.