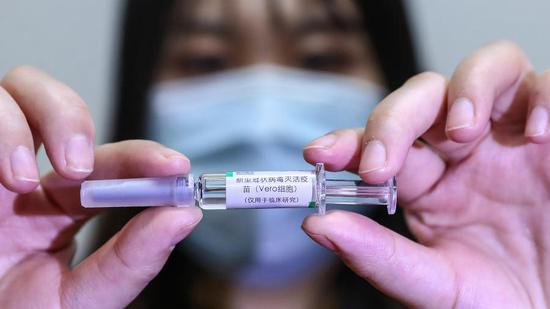
A staff member displays a sample of the COVID-19 inactivated vaccine at a vaccine production plant of China National Pharmaceutical Group Co., Ltd. (Sinopharm) in Beijing, April 10, 2020. /Xinhua
China supports Peru's efforts to arrange a phase 3 clinical trial for a potential COVID-19 vaccine developed by China National Pharmaceutical Group (Sinopharm), Chinese State Councilor and Foreign Minister Wang Yi said on Thursday, adding that the move shows a high degree of trust between the two countries.
In a phone conversation with Peruvian Foreign Minister Mario Lopez, Wang said China understands Peru's need to purchase COVID-19 vaccines. He reiterated China's commitment that its vaccines, once developed and put into use, will be made a global public good and provided to developing countries on a priority basis.
Health authorities of Peru approved the phase 3 clinical trial for the Sinopharm vaccine in August.
Phase 3 trials, which usually involve several thousand participants, allow researchers to gather data on the efficacy and safety of potential vaccines for final regulatory approvals.