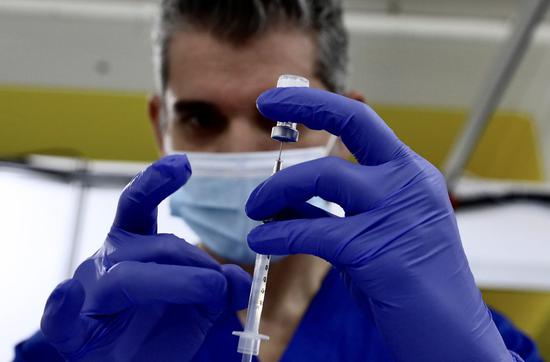
A health care worker prepares a dose of COVID-19 vaccine at a new vaccination site in the California Polytechnic State University in Pomona, Los Angeles County, California, the United States, Feb. 5, 2021. (Xinhua)
American drugmaker Moderna announced Tuesday that the first group of children participants have been vaccinated in its COVID-19 vaccine trial.
The Phase 2/3 study, called the KidCOVE study, will enroll approximately 6,750 pediatric participants in the United States and Canada aged 6 months to less than 12 years.
The trial will evaluate the safety, tolerability, reactogenicity and effectiveness of two doses of Moderna's COVID-19 vaccine mRNA-1273 given 28 days apart, according to the company.
Moderna's COVID-19 vaccine was authorized for emergency use against COVID-19 by the U.S. Food and Drug Administration last December, for individuals 18 years of age and older.